Proangiotensin-12
A Possible Component of the Renin-Angiotensin System


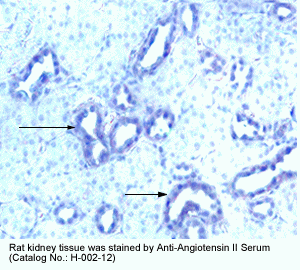
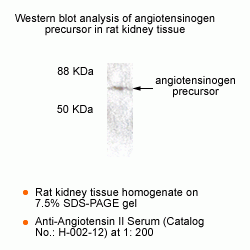
New Anti-Angiotensin I/II (1-7) Antibody for immunohistochemistry
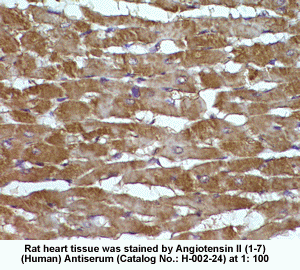 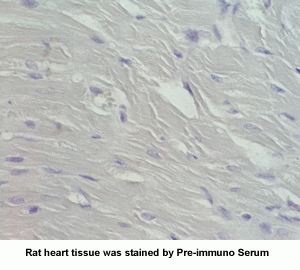
 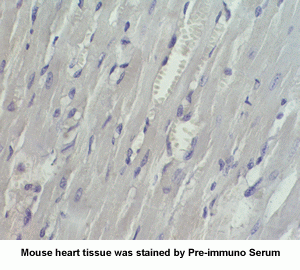
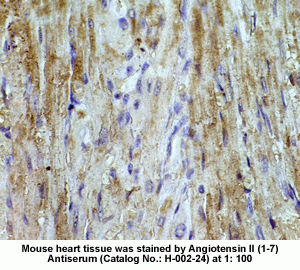 
The renin-angiotensin (RA) system plays an important role in regulating blood pressure and fluid balance. In the search for bioactive peptides with an antibody binding to the N-terminal portion of angiotensin II (Ang II), we isolated a new angiotensinogen-derived peptide from the rat small intestine. Consisting of 12 amino acids, this peptide was termed proangiotensin-12 based on its possible role of an Ang II precursor. Proangiotensin-12 constricted aortic strips and, when infused intravenously, raised blood pressure in rats, while both the vasoconstrictor and pressor response to proangiotensin-12 were abolished by captopril and by CV-11974, an Ang II type I receptor blocker. Proangiotensin-12 is abundant in a wide range of organs and tissues including the small intestine, spleen, kidneys, and liver of rats. The identification of proangiotensin-12 suggests a processing cascade of the RA system, different from the cleavage of angiotensinogen to Ang I by renin.
Nagata S, et al. Biochem Biophys Res Commun. 2006 Oct 5; [Epub ahead of print]
Angiotensin IV (Ang IV) is a metabolite of the potent vasoconstrictor angiotensin II (Ang II). Because specific binding sites for this peptide have been reported in numerous tissues including the brain, it has been suggested that a specific Ang IV receptor (AT4) might exist. Bolus injection of Ang IV in brain ventricles has been implicated in learning, memory, and localized vasodilatation. However, the functions of Ang IV in a physiological context are still unknown. In this study, we generated a transgenic (TG) mouse model that chronically releases Ang IV peptide specifically in the brain. TG mice were found to be hypertensive by the tail-cuff method as compared with control littermates. Treatment with the angiotensin-converting enzyme inhibitor captopril had no effect on blood pressure, but surprisingly treatment with the Ang II AT1 receptor antagonist candesartan normalized the blood pressure despite the fact that the levels of Ang IV in the brains of TG mice were only 4-fold elevated over the normal endogenous level of Ang peptides. Calcium mobilization assays performed on cultured CHO cells chronically transfected with the AT1 receptor confirm that low-dose Ang IV can mobilize calcium via the AT1 receptor only in the presence of Ang II, consistent with an allosteric mechanism. These results suggest that chronic elevation of Ang IV in the brain can induce hypertension that can be treated with angiotensin II AT1 receptor antagonists.
Circ Res. 2004 Jun 11;94(11):1451-7. Epub 2004 Apr 29
Since it has been suggested that angiotensin (Ang) (1-7) functions as an antihypertensive peptide, we studied its effect on the Ang II-enhanced norepinephrine (NE) release evoked by K+ in hypothalami isolated from aortic coarcted hypertensive (CH) rats. The endogenous NE stores were labeled by incubation of the tissues with 3H-NE during 30 min, and after 90 min of washing, they were incubated in Krebs solution containing 25 mM KCl in the absence or presence of the peptides. Ang-(1-7) not only diminished the K+-evoked NE release from hypothalami of CH rats, but also blocked the Ang II-enhanced NE release induced by K+. Ang-(1-7) blocking action on the Ang II response was prevented by [D-Ala7]Ang-(1-7), an Ang-(1-7) specific antagonist, by PD 123319, an AT2-receptor antagonist, and by Hoe 140, a B2 receptor antagonist. Ang-(1-7) inhibitory effect on the Ang II facilitatory effect on K+-stimulated NE release disappeared in the presence of Nomega-nitro-L-arginine methylester and was restored by L-arginine. Our present results suggest that Ang-(1-7) may contribute to blood pressure regulation by blocking Ang II actions on NE release at the central level. This inhibitory effect is a nitric oxide-mediated mechanism involving AT2 receptors and/or Ang-(1-7) specific receptors and local bradykinin generation.
Gironacci MM, et al. Regul Pept. 2004 Apr 15;118(1-2):45-9
The renin-angiotensin system plays a critical role in blood pressure control and body fluid and electrolyte homeostasis. Besides angiotensin (Ang) II, other Ang peptides, such as Ang III [Ang-(2-8)], Ang IV [Ang-(3-8)], and Ang-(1-7) may also have important biological activities. Ang-(1-7) has become an angiotensin of interest in the past few years, because its cardiovascular and baroreflex actions counteract those of Ang II. Unique angiotensin-binding sites specific for this heptapeptide and studies with a selective Ang-(1-7) antagonist indicated the existence of a distinct Ang-(1-7) receptor. We demonstrate that genetic deletion of the G protein-coupled receptor encoded by the Mas protooncogene abolishes the binding of Ang-(1-7) to mouse kidneys. Accordingly, Mas-deficient mice completely lack the antidiuretic action of Ang-(1-7) after an acute water load. Ang-(1-7) binds to Mas-transfected cells and elicits arachidonic acid release. Furthermore, Mas-deficient aortas lose their Ang-(1-7)-induced relaxation response. Collectively, these findings identify Mas as a functional receptor for Ang-(1-7) and provide a clear molecular basis for the physiological actions of this biologically active peptide.
Santos RA, et al. Proc Natl Acad Sci U S A. 2003 Jun 26 [Epub ahead of print]
Central administration of angiotensin IV (Ang IV) or its analogues enhance performance of rats in passive avoidance and spatial memory paradigms. The purpose of this study was to examine the effect of a single bolus injection of two distinct AT4 ligands, Nle1-Ang IV or LVV-haemorphin-7, on spatial learning in the Barnes circular maze. Mean number of days for rats treated with either Nle1-Ang IV or LVV-haemorphin-7 to achieve learner criterion is significantly reduced compared with controls (P < 0.001 and P < 0.05 respectively). This is due to enhanced ability of the peptide-treated rats to adopt a spatial strategy for finding the escape hatch. In all three measures of learning performance, (1) the number of errors made, (2) the distance travelled and (3) the latency in finding the escape hatch, rats treated with either 100 pmol or 1 nmol of Nle1-Ang IV or 100 pmol LVV-haemorphin-7 performed significantly better than the control groups. As early as the first day of testing, the rats treated with the lower dose of Nle1-Ang IV or LVV-haemorphin-7 made fewer errors (P < 0.01 and P< 0.05 respectively) and travelled shorter distances (P < 0.05 for both groups) than the control animals. The enhanced spatial learning induced by Nle1-Ang IV (100 pmol) was attenuated by the co-administration of the AT4 receptor antagonist, divalinal-Ang IV (10 nmol). Thus, administration of AT4 ligands results in an immediate potentiation of learning, which may be associated with facilitation of synaptic transmission and/or enhancement of acetylcholine release.
Lee J, et al. Neuroscience. 2004;124(2):341-9
Angiotensin-(1-7) [Ang-(1-7)] has biological actions that can often be distinguished from those of angiotensin II (Ang II). Recent studies indicate that the effects of Ang-(1-7) are mediated by specific receptor(s). We now report the partial characterization of a new antagonist selective for Ang-(1-7), D-Pro7-Ang-(1-7). D-Pro7-Ang-(1-7) (50 pmol) inhibited the hypertensive effect induced by microinjection of Ang-(1-7) [41 vs 212 mm Hg, 25 pmol Ang-(1-7) alone] into the rostral ventrolateral medulla without changing the effect of Ang II (162.5 vs 192.5 mm Hg after 25 pmol Ang II alone). At 10(-7) mol/L concentration, it completely blocked the endothelium-dependent vasorelaxation produced by Ang-(1-7) (10(-10) to 10(-6) mol/L) in the mouse aorta. The antidiuresis produced by Ang-(1-7) (40 pmol/100 g body weight) in water-loaded rats was also blocked by its analog [1 microg/100 g body weight; 3.080.8 vs 1.270.33 mL in Ang-(1-7)-treated rats]. D-Pro7-Ang-(1-7) at a molar ratio of 40:1 did not change the hypotensive effect of bradykinin. Moreover, D-Pro7-Ang-(1-7) did not affect the dipsogenic effect produced by intracerebroventricular administration of Ang II (11.41.15 vs 8.81.2 mL/h after Ang II) and did not show any demonstrable angiotensin-converting enzyme inhibitory activity in assays with the synthetic substrate Hip-His-Leu and rat plasma as a source of enzyme. Autoradiography studies with 125I-Ang-(1-7) in mouse kidney slices showed that D-Pro7-Ang-(1-7) competed for the binding of Ang-(1-7) to the cortical supramedullary region. In Chinese hamster ovary cells stably transfected with the AT1 receptor subtype, D-Pro7-Ang-(1-7) did not compete for the specific binding of 125I-Ang-II in concentrations up to 10(-6) mol/L. There was also no significant displacement of Ang II binding to angiotensin type 2 receptors in membrane preparations of adrenal medulla. These data indicate that D-Pro7-Ang-(1-7) is a selective antagonist for Ang-(1-7), which can be useful to clarify the functional role of this heptapeptide.
The mammalian brain harbors a renin-angiotensin system (RAS), which is independent from the peripheral RAS. Angiotensin II is a well-studied member of the RAS and exerts most of the known angiotensin-mediated effects on fluid and electrolyte homeostasis, autonomic activity, neuroendocrine regulation, and behavior. This review summarizes a mass of compelling new evidence for the biological role of an active (3-8) fragment of angiotensin II, named angiotensin IV. Angiotensin IV binds to a widely distributed binding site in the brain, but which is different from the known angiotensin II receptors AT1 and AT2. Angiotensin IV has been implicated in a number of physiological actions, including the regulation of blood flow, the modulation of exploratory behavior, and processes attributed to learning and memory. Furthermore, angiotensin IV may also be involved in neuronal development. Collectively, the available evidence suggests that angiotensin IV is a potent neuropeptide, involved in a broad range of brain functions.
Von Bohlen Und Halbach O. Cell Tissue Res 2003 Jan;311(1):1-9
Although angiotensin IV (Ang IV) was thought initially to be an inactive product of Ang II degradation, it was subsequently shown that the hexapeptide markedly enhances learning and memory in normal rodents and reverses the memory deficits seen in animal models of amnesia. These central nervous system effects of Ang IV are mediated by binding to a specific site, known as the AT(4) receptor, which is found in appreciable levels throughout the brain and is concentrated particularly in regions involved in cognition. This field of research was redefined by the identification of the AT(4) receptor as the transmembrane enzyme, insulin-regulated membrane aminopeptidase (IRAP). Here, we explore the potential mechanisms by which Ang IV binding to IRAP leads to the facilitation of learning and memory.
Albiston AL, et al. Trends Endocrinol Metab 2003 Mar;14(2):72-7
Although angiotensin II has long been considered to represent the end product of the renin-angiotensin system (RAS), there is accumulating evidence that it encompasses additional effector peptides with diverse functions. In this respect, angiotensin IV (Ang IV) formed by deletion of the two N terminal amino acids, has sparked great interest because of its wide range of physiological effects. Among those, its facilitatory role in memory acquisition and retrieval is of special therapeutic relevance. High affinity binding sites for this peptide have been denoted as AT(4)- receptors and, very recently, they have been proposed to correspond to the membrane-associated OTase/ IRAP aminopeptidase. This offers new opportunities for examining physiological roles of Ang IV in the fields of cognition, cardiovascular and renal metabolism and pathophysiological conditions like diabetes and hypertension. Still new recognition sites may be unveiled for this and other angiotensin fragments. Recognition sites for Ang-(1-7) (deletion of the C terminal amino acid) are still elusive and some of the actions of angiotensin III (deletion of the N terminal amino acid) in the CNS are hard to explain on the basis of their interaction with AT(1)-receptors only. A more thorough cross-talk between in vitro investigations on native and transfected cell lines and in vivo investigations on healthy, diseased and transgenic animals may prove to be essential to further unravel the molecular basis of the physiological actions of these small endogenous angiotensin fragments.
Vauquelin G, et al. J Renin Angiotensin Aldosterone Syst 2002 Dec;3(4):195-204
The role of angiotensin IV (AngIV) in the regulation of angiotensin-converting enzyme (ACE) was studied in vitro. This study demonstrates that this active fragment appeared as a novel endogenous ACE inhibitor. Inhibitory kinetic studies revealed that AngIV acts as a purely competitive inhibitor with a K(i) value of 35 microM. AngIV was found to be quite resistant to ACE hydrolysis opposite to hemorphins which are both ACE inhibitors and substrates. In order to confirm a putative role of AngIV and hemorphins in the Renin-Angiotensin system (RAS) regulation, we studied their influence on AngI conversion. We noticed that 16.7 microM of both peptides decreased more than 50% of AngI conversion to AngII in vitro. The capacity of hemorphins, particularly LVVH-7, and AngIV to inhibit ACE activity here suggests a synergistic relation between these two peptides and the regulation of RAS.
Fruitier-Arnaudin I, et al. Peptides 2002 Aug;23(8):1465-70
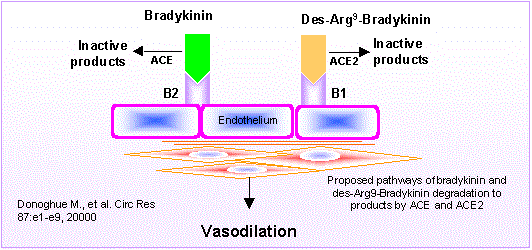
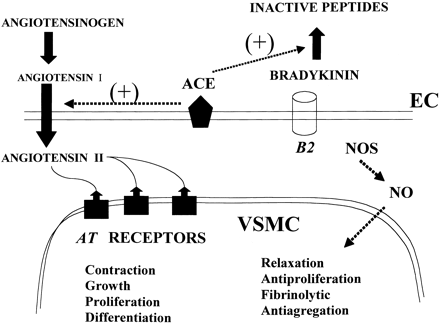
Biosynthetic pathways for the formation of neuroactive peptides and the processes for their inactivation include several enzymatic steps. In addition to enzymatic processing and degradation, several neuropeptides have been shown to undergo enzymatic conversion to fragments with retained or modified biological activity. This has most clearly been demonstrated for e.g. opioid peptides, tachykinins, calcitonin gene-related peptide (CGRP) as well as for peptides belonging to the renin-angiotensin system. Sometimes the released fragment shares the activity of the parent compound. However, in many cases the conversion reaction is linked to a change in the receptor activation profile, i.e. the generated fragment acts on and stimulates a receptor not recognized by the parent peptide. This review will describe the characteristics of certain neuropeptide fragments having the ability to modify the biological action of the peptide from which they are derived. Focus will be directed to the tachykinins, the opioid peptides, angiotensins as well as to CGRP, bradykinin and nociceptin. The kappa opioid receptor selective opioid peptide, dynorphin, recognized for its ability to produce dysphoria, is converted to the delta opioid receptor agonist Leu-enkephalin, with euphoric properties. The tachykinins, typified by substance P (SP), is converted to the bioactive fragment SP(1-7), a heptapeptide mimicking some but opposing other effects of the parent peptide. The bioactive angiotensin II, known to bind to and stimulate the AT-1 and AT-2 receptors, is converted to angiotensin IV (i.e. angiotensin 3-8) with preference for the AT-4 sites or to angiotensin (1-7), not recognized by any of these receptors. Both angiotensin IV and angiotensin (1-7) are biologically active. For example angiotensin (1-7) retains some of the actions ascribed for angiotensin II but is shown to counteract others.Thus, it is obvious that the activity of many neuroactive peptides is modulated by bioactive fragments, which are formed by the action of a variety of peptidases. This phenomenon appears to represent an important regulatory mechanism that modulates many neuropeptide systems but is generally not acknowledged.
Hallberg M, Nyberg F. Curr Protein Pept Sci 2003 Feb;4(1):31-44
The role of angiotensin II (AII) and angiotensin IV (AIV) as inducers of PAI-1 expression during hypertension was studied in vivo. A 2-week infusion of AII (300 ng/kg/min) via an osmotic pump increased systolic blood pressure (171 2 vs. 138 6 mm Hg), urinary protein excretion (32 6 vs. 14 2 mg/day), and renal (2.2 0.5 vs. 1.0 0.1) and cardiac (1.8 0.3 vs. 1.0 0.1) gene expression of plasminogen activator inhibitor 1 (PAI-1). AIV infusion did not affect any of the above with the exception of PAI-1 gene expression which was increased in the left ventricles (1.7 0.3 vs. 1.0 0.1). AII-infused rats displayed a decreased creatinine clearance (538 75 vs. 898 96 ml/min) and hypertrophic left ventricles (0.275 0.006 vs. 0.220 0.011 g/100 g). Our results demonstrate that AII but not AIV infusion is associated with increased renal PAI-1 gene expression.
Abrahamsen CT, et al. Pharmacology 2002 Sep;66(1):26-30
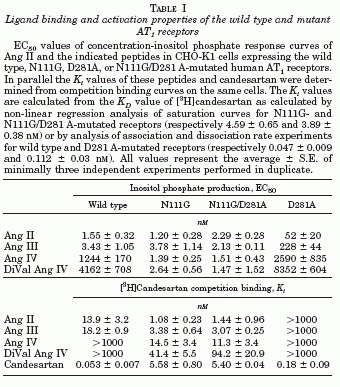
The octapeptide hormone, angiotensin II (Ang II), exerts its major physiological effects by activating AT(1) receptors. In vivo Ang II is degraded to bioactive peptides, including Ang III (angiotensin-(2-8)) and Ang IV (angiotensin-(3-8)). These peptides stimulate inositol phosphate generation in human AT(1) receptor expressing CHO-K1 cells, but the potency of Ang IV is very low. Substitution of Asn(111) with glycine, which is known to cause constitutive receptor activation by disrupting its interaction with the seventh transmembrane helix (TM VII), selectively increased the potency of Ang IV (900-fold) and angiotensin-(4-8), and leads to partial agonism of angiotensin-(5-8). Consistent with the need for the interaction between Arg(2) of Ang II and Ang III with Asp(281), substitution of this residue with alanine (D281A) decreased the peptide's potency without affecting that of Ang IV. All effects of the D281A mutation were superseded by the N111G mutation. The increased affinity of Ang IV to the N111G mutant was also demonstrated by binding studies. A model is proposed in which the Arg(2)-Asp(281) interaction causes a conformational change in TM VII of the receptor, which, similar to the N111G mutation, eliminates the constraining intramolecular interaction between Asn(111) and TM VII. The receptor adopts a more relaxed conformation, allowing the binding of the C-terminal five residues of Ang II that switches this "preactivated" receptor into the fully active conformation. Le MT, et al. J Biol Chem 2002 Jun 28;277(26):23107-10
OBJECTIVE: The aim of the present study was to investigate whether angiotensin II (Ang II), angiotensin III (Ang III) or Ang II (2-8), angiotensin IV (Ang IV) or Ang II (3-8) and Ang II (1-7), Ang II (4-8), Ang II (5-8) and Ang II (1-4) can stimulate collagen gel contraction in cardiac fibroblasts in serum-free conditions. METHODS: Cardiac fibroblasts (from male adult Wistar rats) from passage 2 were cultured to confluency and added to a hydrated collagen gel in a Dulbecco's Modified Eagle's Medium, with or without foetal bovine serum, for one, two or three days. The area of the collagen gels embedded with cardiac fibroblasts was determined by a densitometric analysis. Collagen gel contraction was characterised by a decrease in the gel area. RESULTS: Ang II dose-dependently stimulated the contraction of collagen mediated by cardiac fibroblasts after one, two or three days of incubation in a serum-free medium. Telmisartan completely blocked the Ang II-induced collagen contraction by cardiac fibroblasts. PD 123319 and des-Asp(1)-Ile(8)-Ang II had no effect on the Ang II-induced collagen contraction by cardiac fibroblasts. Ang III also stimulated the contraction of collagen mediated by cardiac fibroblasts after one, two or three days of incubation in a serum-free medium. des-Asp(1)-Ile(8)-Ang II and telmisartan completely blocked the Ang III-induced collagen gel contraction by cardiac fibroblasts. des-Asp(1)-Ile(8)-Ang II, however, had no effect on the Ang II-induced collagen gel contraction by cardiac fibroblasts. Ang IV and Ang II (4-8), (5-8), (1-7) and (1-4), however, had no effect on collagen gel contraction by cardiac fibroblasts. Addition of telmisartan, PD 123319 or des-Asp(1)-Ile(8)-Ang II alone did not affect collagen gel contraction by cardiac fibroblasts. CONCLUSION: Our data demonstrate that the effects of Ang II on the collagen gel contraction by adult rat cardiac fibroblasts in serum-free conditions are Ang II type 1(AT(1))-receptor- mediated, because they are abolished by the specific AT(1)-receptor antagonist, telmisartan, and not by the AT(2)-receptor antagonist PD 123319 or by the Ang III antagonist des-Asp(1)-Ile(8)-angiotensin. The Ang III- stimulated contraction of collagen by cardiac fibroblasts is completely blocked by the Ang III receptor antagonist, des-Asp(1)-Ile(8)-angiotensin II, and by telmisartan.
Lijnen P, et al. J Renin Angiotensin Aldosterone Syst 2002 Sep;3(3):160-6
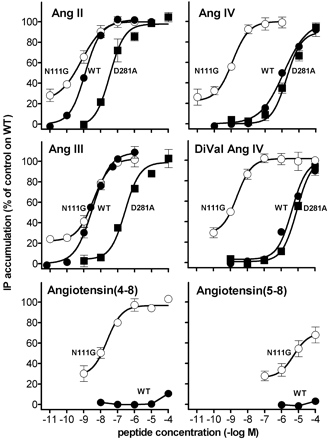
Activation of AT1 receptor mutants by angiotensin-related peptides. IP production mediated by incubation with Ang II and the indicated peptides in CHO-K1 cells expressing the wild type (filled circles), N111G (open circles), or D281A (filled squares) mutated human AT1 receptors are shown. Each point represents the average of minimally four independent experiments performed in duplicate and is expressed as percent of the maximal response of Ang II (1 µM) on
wild type AT1 receptor-expressing cells.
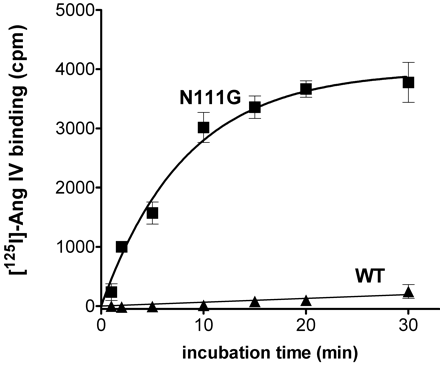
Specific binding of [125I]Ang IV to wild type and N111G mutant AT1 receptors. CHO-K1 cells expressing wild type (filled triangles) or N111G (filled squares) mutated human AT1 receptors were incubated with 0.5 nM [125I]Ang IV at 37 °C for the indicated periods of time. Specific binding was calculated by subtracting nonspecific binding in the presence of 1 µM unlabeled Ang IV.
|


